Hundreds more children suffering from rare muscle-wasting condition spinal muscular atrophy (SMA) are set to gain access to “life-changing” treatments following their approval for widespread use across the NHS.
One mother, whose son benefited from an early access scheme for one of these therapies, attested that it had “fundamentally changed the course of his life”.
The injectable drug nurinersen, which marks the first disease-modifying therapy for SMA, alongside the oral treatment risdiplam, were previously available through a special access programme while further evidence was gathered.
Now, the National Institute for Health and Care Excellence (Nice) has given its endorsement for the routine and widespread application of these treatments within the NHS in England.
In its final draft guidance, Nice highlighted that the drugs have the potential to improve survival rates, slow the progression of the disease, and help individuals maintain their independence.
SMA is a genetic condition which causes severe muscle weakness and can affect breathing, swallowing and movement.
Without treatment, the disease can lead to profound disability and reduced life expectancy – babies with the most severe form of the disease are not expected to live beyond two years.
Nusinersen, also called Spinraza and made by Biogen, is given via periodic spinal injection while risdiplam, also known as Evrysdi and made by Roche, is a daily oral medicine which can be given as syrup or by a pill.
NHS England said the therapies have now helped 73 children with a severe form of SMA (Type 1) survive to aged five or older.
Ezra Thorman, now nine, started treatment with nusinersen when he was five-months old through an NHS access programme.
He was diagnosed with SMA Type 1 in 2016 and is now in year 4 at school in Ramsgate, Kent.
His mother Portia Thorman said: “Starting mainstream school is a milestone we were once told Ezra would never reach.
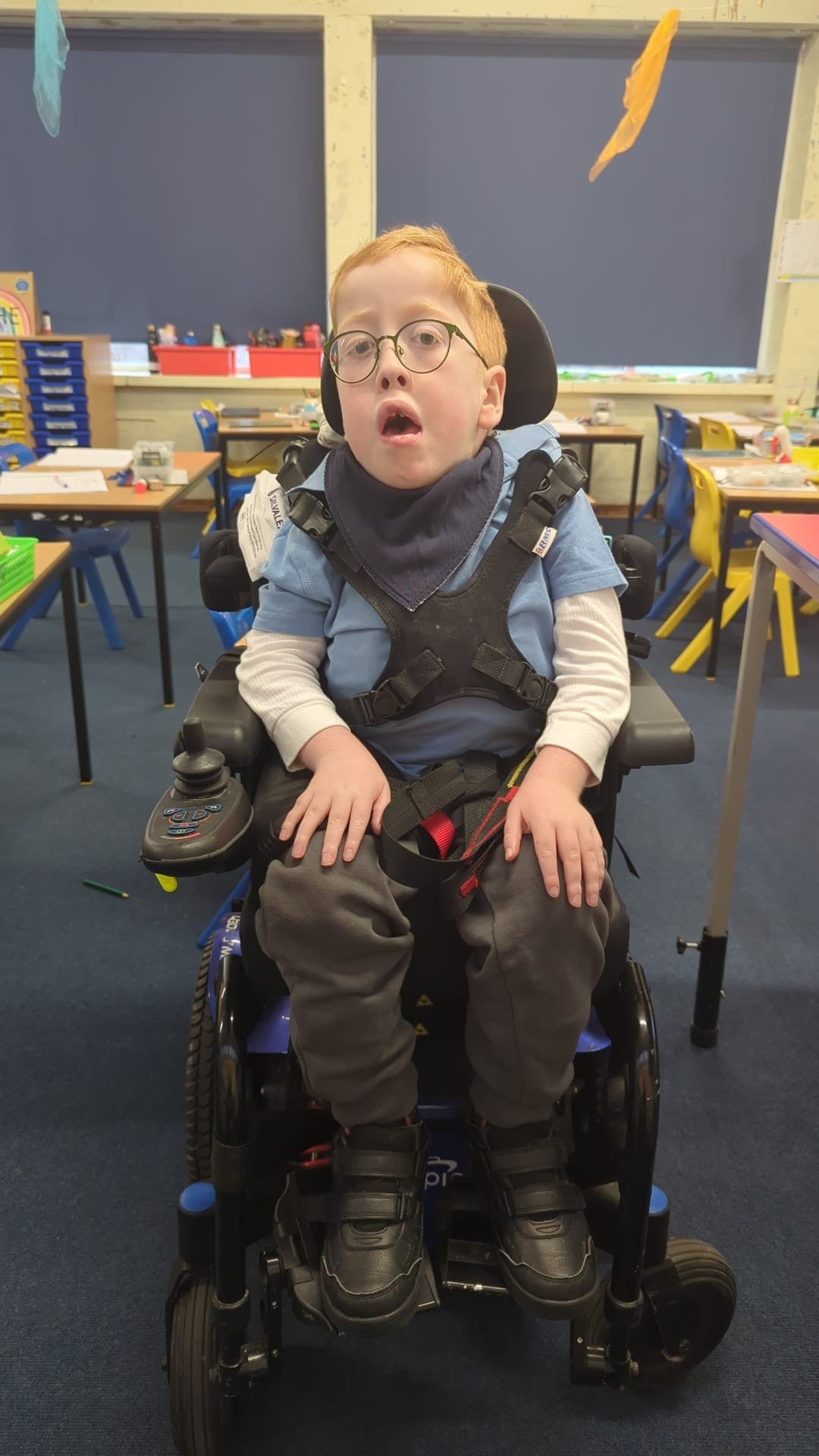
“His SMA Type 1 diagnosis in 2016 came with a life expectancy of under two-years-old.
“The hope for a future this bright felt out of reach, however accessing treatment at just five months old fundamentally changed the course of his life.”
Around 70 babies are born with SMA each year in the UK and there are around 1,150 people living with the condition in England.
Data from an SMA database show more than 350 people with SMA receiving treatment are aged five and over – 107 of whom have been treated with nusinersen, 200 with risdiplam and 45 with gene therapy Zolgensma, which is also routinely offered by the NHS in England.
Professor James Palmer, national medical director for specialised services at NHS England, said: “These lifeline treatments have offered a phenomenal step forward in care for children and families affected by such a debilitating condition, and it is fantastic that they will now be available on the NHS in the long-term.
“For parents who faced the unimaginable pain of thinking their child would not reach their second birthday, they now have hope of seeing them walk to school and play with their friends, thanks to these life-changing new therapies on the NHS.”
Helen Knight, director of medicines evaluation at Nice, said: “After carefully reviewing the latest evidence and real-world experience from the NHS, our independent committee concluded that nusinersen and risdiplam can offer substantial, life-changing benefits for many people with SMA.

“These treatments can help people live longer, maintain their independence, communication and participation in everyday life, and reduce the need for hospital care.”
Public health minister Sharon Hodgson said: “Drawing on both clinical evidence and the real-world experiences of patients already receiving treatment, this decision will help ensure more people with SMA can access the care and support they need to live fuller, more independent lives.”
Giles Lomax, chief executive of SMA UK, said: “This is a historic moment for the SMA community and follows many years of determined campaigning by patients, families, clinicians, charities and wider stakeholders to secure long-term access to these life-changing treatments.
“After years of uncertainty, people can finally feel confident that nusinersen and risdiplam will remain available on the NHS.”
It comes after it was announced that around 750,000 newborns in England will be checked for SMA as part of a new study to assess whether screening should be included in post-birth baby checks for serious health conditions.
Pop star Jesy Nelson has campaigned for screening to be rolled out after her twins were diagnosed with the condition.
Scientists at the University of Oxford are now to assess the feasibility, acceptability, effectiveness and cost-effectiveness of adding SMA to the heel-prick blood test given to newborns.
Nelson’s twins, Ocean Jade and Story Monroe Nelson, were diagnosed with the genetic condition, which causes progressive muscle wastage, movement problems, issues with breathing and swallowing, muscle tremors, as well as bone and joint problems.
The singer has since dedicated herself to raising awareness of SMA and urged the Government to add the condition to the newborn blood spot screening test, as early treatment can help avoid some of its most devastating effects.



